|
11/7/2022 0 Comments Atomic radius across a period 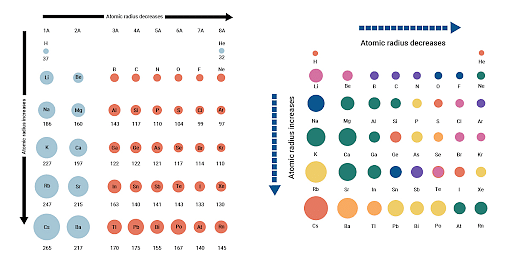
Secondly, since the electron cloud surrounding the atom does not have a sharp boundary, the determination of the atomic size cannot be precise. It does not store any personal data.The size of an atom is very small (120 pm). The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The added electron is slightly repelled by the other electrons in the shell, while the added proton increases the positive charge of the nucleus. How does an atom change from left to right on the periodic table?Īs you go across a period from left to right and atom to atom, each step adds another electron to the same energy level (shell) and a proton to the nucleus. 
Effective nuclear charge decreases because the inner electrons repel the outer electrons, weakening the nucleus pull for the outer electrons. The size increase because the effective nuclear charge (positive charge of nucleus) experienced by the outer electrons decreases down a group. This increase in nuclear charge cause the nucleus to attract the electrons and their orbitals more strongly, shrinking the size of the atom. What causes the size of an atom to decrease? As a result, the atomic radius decreases. The valence electrons are held closer towards the nucleus of the atom. This is because, within a period or family of elements, all electrons are added to the same shell. Why does atomic radius decrease across a period?Ītomic size gradually decreases from left to right across a period of elements. Ionization energy increases from left to right in a period and decreases from top to bottom in a group. Moving left to right within a period or upward within a group, the first ionization energy generally increases. How does ionization energy vary in periods and groups? 
Out of these elements, sodium is the largest in size due to the presence of three shells whereas lithium has two and hydrogen has one shell only. The largest elements in group 1, 2 and 3 are helium, lithium and sodium respectively. A higher effective nuclear charge causes greater attractions to the electrons, pulling the electron cloud closer to the nucleus which results in a smaller atomic radius. How does atomic size increase in a period?Īcross a period, effective nuclear charge increases as electron shielding remains constant. Z, and shielding by other electrons, underlies the structure of the Periodic Table. This contest between nuclear charge, i.e. Atomic radii thus INCREASE down the Group. This results in a DECREASE in atomic radii across the Period, due to the increased nuclear charge which draws in the valence electrons. Why does atomic size increase down a group but decrease across a period? Since the force of attraction between nuclei and electrons increases, the size of the atoms decreases. These electrons are gradually pulled closer to the nucleus because of its increased positive charge. Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level. Why does atomic size decrease in a period?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
